RR6 Model
RR6 Calculator
The RR6 model predicts survival in myelofibrosis based on clinical response after 6 months of ruxolitinib.
Reference
Maffioli M, Mora B, Ball S, Iurlo A, Elli EM, Finazzi MC, Polverelli N, Rumi E, Caramella M, Carraro MC, D'Adda M, Molteni A, Sissa C, Lunghi F, Vismara A, Ubezio M, Guidetti A, Caberlon S, Anghilieri M, Komrokji RS, Cattaneo D, Della Porta MG, Giorgino T, Bertu L, Brociner M, Kuykendall AT, Passamonti F. A Prognostic Model to Predict Survival After 6 Months of Ruxolitinib in Patients with Myelofibrosis. Blood Advances 2022. bloodadvances.2021006889. doi:10.1182/bloodadvances.2021006889
IMPORTANT: This tool is for educational use only. It does not constitute medical advice. It should not be used for medical diagnosis and/or medical treatment.
RR6 model results
- Risk stratum
- IR - Intermediate
- Median overall survival*
- 61 months
- Median o. survival* 95% CI
- 43-80 months
*counted from 6 months post ruxolitinib start.
Calculation
Risk points:
- spleen length reduction <=30%: 0
- low ruxolitinib dose: 1
- transfusion status: 0
Total risk points: 1
Legend
LCM - left costal margin
BID - bis in die, twice daily
RBC - red blood cell
Survival curves
Actuarial survival curves of the 3 risk groups of patients according to the Response to Ruxolitinib after 6 months (RR6) developed in ruxolitinib-treated myelofibrosis patients (training cohort).
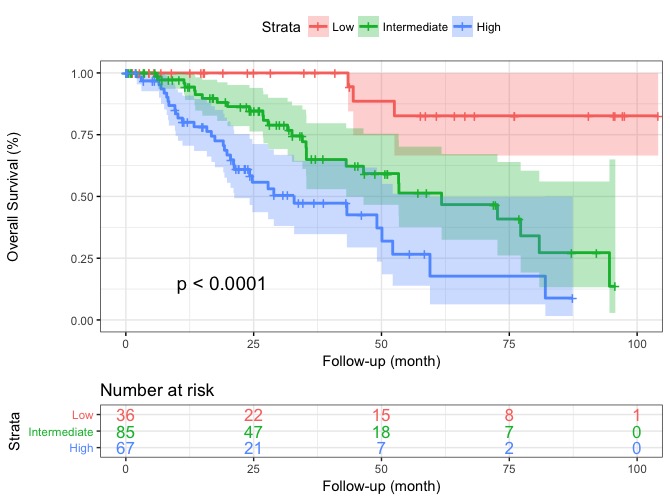
Source: Maffioli et al., A Prognostic Model to Predict Survival After 6 Months of Ruxolitinib in Patients with Myelofibrosis. Blood Advances. doi:10.1182/bloodadvances.2021006889